The Millennium Cohort Study (MCS) is a nationally representative cohort of children born between September 2000 and January 2002 in the United Kingdom23. Households with a child aged 9 months of age (Sweep 1) and living in the UK were randomly selected, providing a nationally representative sample23. Smaller UK nations, and in England disadvantaged areas, and families with ethnic minority backgrounds were oversampled, and at age 3 (Sweep 2), further eligible children—not captured at baseline—were added to the sample. Participants have been followed up seven times to date: at the average age of 9 months (Sweep 1: 2000–2002), 3 years (Sweep 2: 2003–2005), 5 years (Sweep 3: 2006), 7 years (Sweep 4: 2008), 11 years (Sweep 5: 2012–2013), 14 years (Sweep 6: 2015–2016), and 17 years (Sweep 7: 2017–2019). The total sample size comprises 19,519 children within 19,244 families ever interviewed24. In the analytical sample, we included children if they participated in the age 17 sweep and did not have missing values: data on self-reported general health covered all four UK nations; hospital episodes were only available for England. All methods were carried out in accordance with relevant guidelines and regulations and informed consent was obtain from all subjects.
Exposure to air pollution
Annual average PM2.5, PM10, and NO2 were estimated for Europe using Geographically and Temporally Weighted Regression (GTWR) between 2000 and 2019 on a 25 × 25 m grid. Detailed descriptions of the model development can be found in Shen et al.25. Briefly, the GTWR models regressed annual average observations from routine monitoring stations on several spatial predictor variables, such as chemical transport model estimates, satellite-derived data, meteorological data, and land-use and road variables, capturing spatio-temporal variations in the measured annual average air pollution levels. GTWR allowed the regression coefficients to vary in space and time, reflecting the changing dynamics of air pollution across Europe over a 20-year period. The GTWR models showed satisfactory performance, explaining 71–82% of the variance in PM2.5 levels, 50–68% of the variance in PM10 levels and 61–69% of the variance in NO2 levels, as given by 5-fold cross-validated R2 values25.
Residential postcodes at interviews—as well as at birth for those moving between birth and sweep 1—were geocoded into 1-m resolution grid reference (i.e. easting [X], northing [Y]) using the British National Grid (England, Scotland, and Wales) and the Irish Grid (Northern Ireland) coordinate systems. Postcodes are the smallest geographic units in the UK, there are approximately 1.8 million live postcodes with an average of 18 households each. Monthly residential postcode history was created using information about the dates of birth, dates of interviews, and residential moves. If the interview date was missing despite a productive interview, country-specific median dates were imputed. Main respondents were asked in each sweep whether their address had changed since the last interview, and if so, when they moved to their current address; to avoid discrepancies, we considered move dates valid if they took place between two productive interviews.
Average residential air pollution exposure for each year of participants’ lives were derived using buffers around postcode centroids. Monthly postcode history was linked to annual air pollution concentrations and averaged for every 12 months from birth onwards. Instead of extracting the pollution concentration at the grid reference, we computed 100, 200, and 500-m buffers around postcode centroids and extracted the area-weighted average pollution concentration within buffers. Using buffers aimed to lower exposure misclassification due to distance error between postcode and property centroids. As previous internal CLS analyses on MCS COVID web survey data suggested that the mean distance error between postcode and residential centroid is 60 m (SD = 87 m)26, we chose 200-m buffers as the main analyses as it likely covered at least 95% of the residential properties. Finally, we computed mean exposure during infancy (0–1 years), early childhood (2–4 years), middle childhood (5–7 years), late childhood (8–11 years), and adolescence (from 12 years until the interview in Sweep 7 [~ 17 years]), as well as during participants’ whole life (i.e., accumulated exposure, 0–17 years); classification aligns with schooling milestones in the UK (i.e. pre-school, key stage 1–4). Supplementary Fig. 1 suggested that the exposure specific correlations across life years, as well as correlations between developmental period-specific PM2.5, PM10 and NO2 exposures were very high among participants.
Outcomes
General health was measured using a self-reported and a health administrative data derived variable.
Self-reported general health
MCS participants at Sweep 7 (average age of 17) were asked how they would describe their health generally, with five response options provided (‘Excellent’; ‘Very Good’; ‘Good’; ‘Fair’; and ‘Poor’). As there were very few individuals with ‘Poor’ and ‘Fair’ general health, we merged these groups together.
Number of hospital episodes
At Sweep 7, participants were asked to provide written consent linking their health records to the survey data27, and 85% of the sample provided consent. For MCS participants residing in England, extraction from the Hospital Episode Statistics (HES) database was carried out by National Health Service (NHS) England using information on name, sex, date of birth, and postcode27,28. The match rate was 81.5%; however, for participants not linked to HES records we assumed the absence of hospital attendances, rather than the failure of linkage processes29. Hospital episodes (i.e., continuous period of care under one consultant) were extracted from the Admitted Patient Care (HES-APC) dataset which covers almost the entire study period for consented and linked participants (01.01.2001–31.03.2020) and provides information about emergency and non-emergency admissions to secondary care requiring a hospital bed30. HES-APC has an almost universal coverage with 99% of hospital activity in England being funded by NHS30. We derived the number of hospital episodes from early childhood (2 year), middle childhood (5 year), late childhood (8 year), adolescence (12 year) and young adulthood (18 year) onwards as outcomes.
Covariates
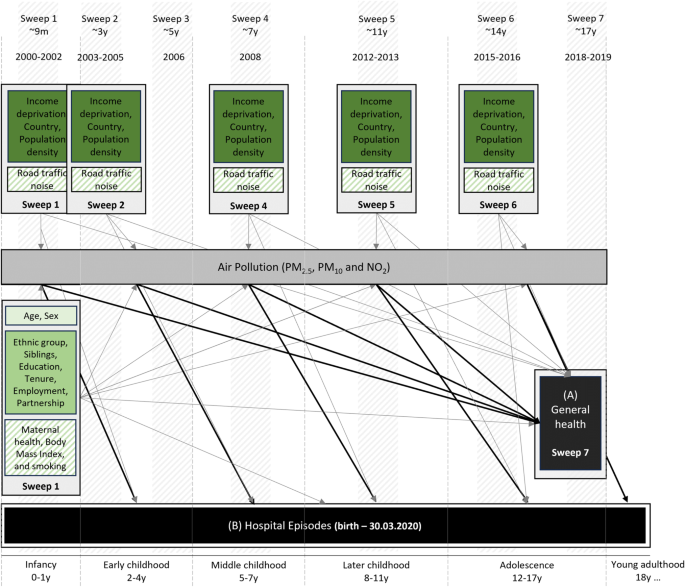
Confounders were selected using a directed acyclic graph, taking into consideration the time of exposures (Fig. 1). Individual-level covariates were time-invariant and derived at 9 months; if not available, at age 3. They included sex (male; female), ethnic groups (white; non-white [specific ethnic groups were merged to allow testing effect modification]), number of siblings (none; 1; 2 or more), parental partnership status (single parent; living with partner), household employment, household tenure, and highest household education. Household employment was derived from the main caregiver and their partners employment status (both employed; one employed; none employed), for household tenure ‘own’ (outright, with mortgage/loan, shared equity), ‘social rent’, ‘private rent’, and ‘other’ (e.g. living with parents, squatting) options were considered. Highest household education was defined based on the National Vocational Qualification (NVQ) scale: the highest level between the main caregiver and their partner was taken and merged into four groups: ‘None or overseas only’, ‘NVQ 1–2’, ‘NVQ 3’ and ‘NVQ 4–5’. We extracted age (in years) at Sweep 7 to account for small differences in the time of outcome data collection.
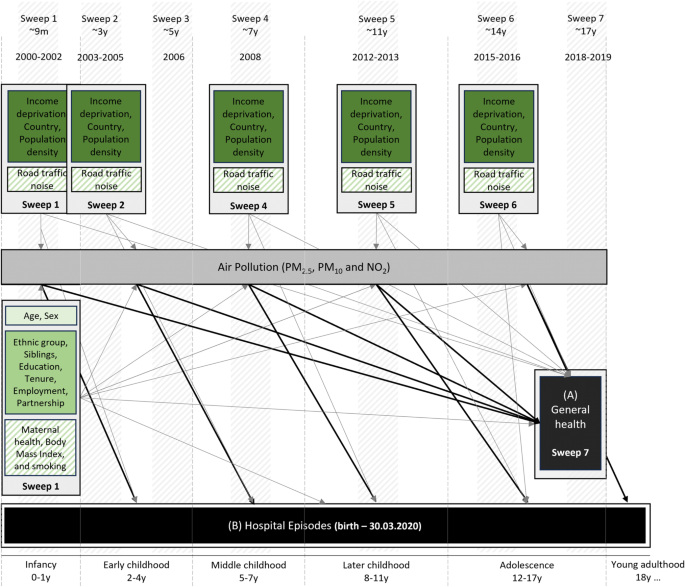
Graph showing assessed life-course associations between air pollution and general health. Black arrows are associations of interest, grey arrows are confounding pathways. Light-green shaded box includes Model 1, medium-green box Model 2, and dark-green box Model 3 confounders; boxes with diagonal stripes are sensitivity analyses. Data collection sweeps and developmental periods are also shown.
Area-level confounders at the time of air pollution exposure were entered in the models as time-variant. They were linked to sweep addresses, and included country (England, Northern Ireland, Scotland, and Wales), population density and income deprivation (see Supplementary Table 1 for more detail). Population density and income deprivation was measured across small areas: lower layer super output areas for England and Wales (1000 to 3000 persons), data zones for Scotland (500 to 1000 persons), and super output areas for Northern Ireland (1300 to 2800 persons). Population density (population counts per hectare) was derived from the 2001 and 2011 Census estimates. Small area-level relative deprivation is published independently across the four nations as part of English, Northern Irish, Scottish and Welsh indices of multiple deprivation; the income deprivation domain was selected as most consistently measured across nations and time. Country-specific income deprivation ranks were classified into four equal categories ranging from ‘Q1—Most deprived’ to ‘Q4—Least deprived’.
Statistical analysis
Analyses for self-reported general health were conducted using proportional odds models, considering the stepwise increasing ordinal nature of the outcome. For the number of hospital episodes, we conducted quasi-Poisson regression to avoid overdispersion. MCS was designed to be representative of the UK population: to account for oversampling as well as to restore the representativeness we applied UK-wide (self-reported general health) and England-only (hospital episodes) complex survey weights in all presented analyses, including descriptive statistics. Analytical samples were separately defined for each exposure period to avoid restriction to children participating in all sweeps; analyses were run separately for each air pollutant.
In Model 1, we adjusted for age and sex. In Model 2, all individual-level variables were added (i.e., age, sex, ethnic groups, number of siblings, partnership status, highest household education, household tenure, and household employment). Finally, in Model 3, we additionally included population density, area income deprivation, as well as country at the time of exposure. Odds Ratios (ORs) or Incidence Rate Ratios (IRRs) and their 95% Confidence Intervals (95% CI) are reported per 1-µg m−3 increment; we also provide coefficients expressed as interquartile range (IQR) change. For self-reported general health at age 17, we run a post-hoc analysis estimating the associations for each year of exposure, and plotted ORs with smoothed curves using LOESS regression. Effect modification by sex, ethnic groups, highest household education and area-level income deprivation at the time of exposure was tested by adding an interaction term to Model 3. As we tested a very large number of interactions (i.e. by three pollutants and two outcomes), only false discovery rate (FDR) adjusted significant (pFDR) findings are interpreted to lower the risk of false positives due to multiple comparisons31.
Six sensitivity and supplementary analyses were carried out to test the robustness of our findings. First, we presented main results after deriving air pollution exposure using 100-m and 500-m buffers (S1). Second, fully-adjusted models were further adjusted for covariates about mother’s health status: self-reported general health (excellent; good; fair; poor), Body Mass Index before birth (underweight [< 18.5]; normal weight [18.5–24.9]; overweight [25.0–29.9]; obese [≥ 30]), and smoking status (never smoked; smoked during pregnancy; reduced or quit smoking before pregnancy; smoked before or after pregnancy) (S2). Third, road traffic noise (Lden) was added to the final models: we downloaded the 2012 noise maps separately for each nations for (a) major roads (≥ 3,000,000 vehicles per year) and (b) road noise in agglomerations (≥ 100,000 residents). Maps were intersected with residential postcode centroids and extracted values were classified into 3 groups (≤ 54.9 decibel [dB]; 55.00–59.9 dB; ≥ 60 dB) (S3). Fourth, to account for bias due to missing individual and area-level confounders (8–9%) we ran multiple imputations on 25 datasets and combined coefficients using Rubin’s rule. In addition to all study variables, auxiliary variables were added including income quartiles, longstanding illness, maternal diabetes, sweep when family entered the study (sweep 1 or sweep 2), governmental region, as well as time-varying urban/rural classification and overall deprivation (S4). Fifth, one of the main underlying assumptions of proportional odds regression is proportional effects of exposure on outcome thresholds, thus we also provided fully adjusted models for self-reported general health using multinomial regression (S5). Finally, as two-pollutant models were not feasible given high multicollinearity, we provided estimates for PM10, PM2.5 and NO2 mixtures using quantile g-computation for multinomial outcomes32 for self-reported general health (S6).
Analyses were conducted in R version 4.3.033, using the survey34, svyVGAM35, and mice36 packages.
link